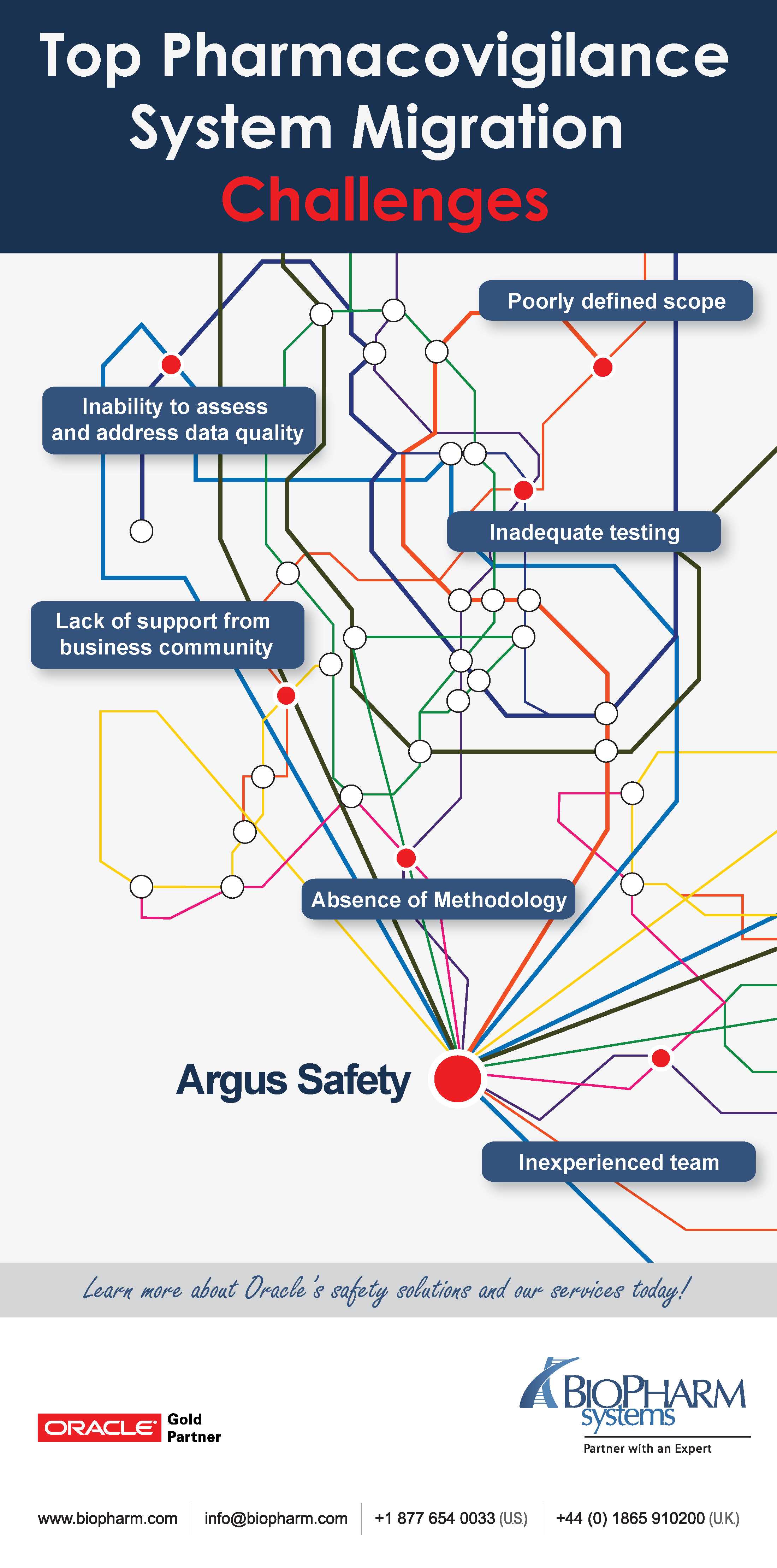
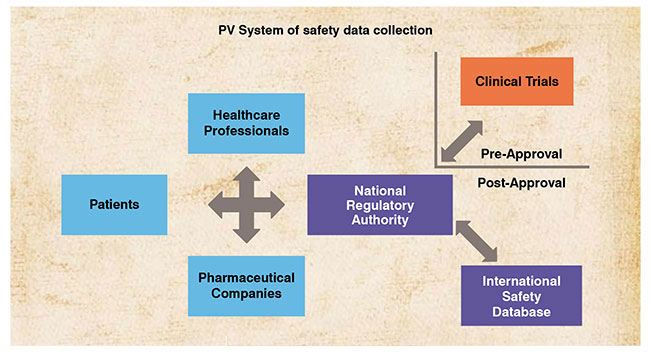
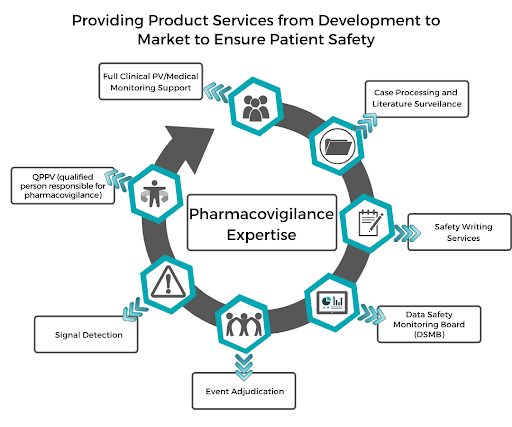
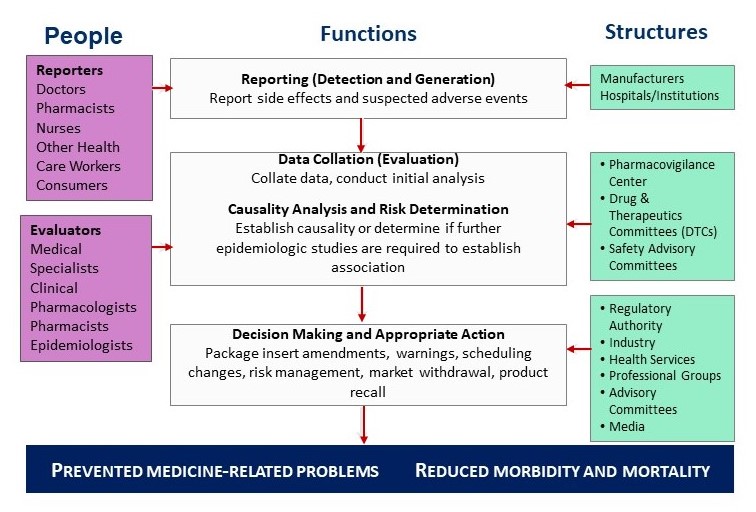
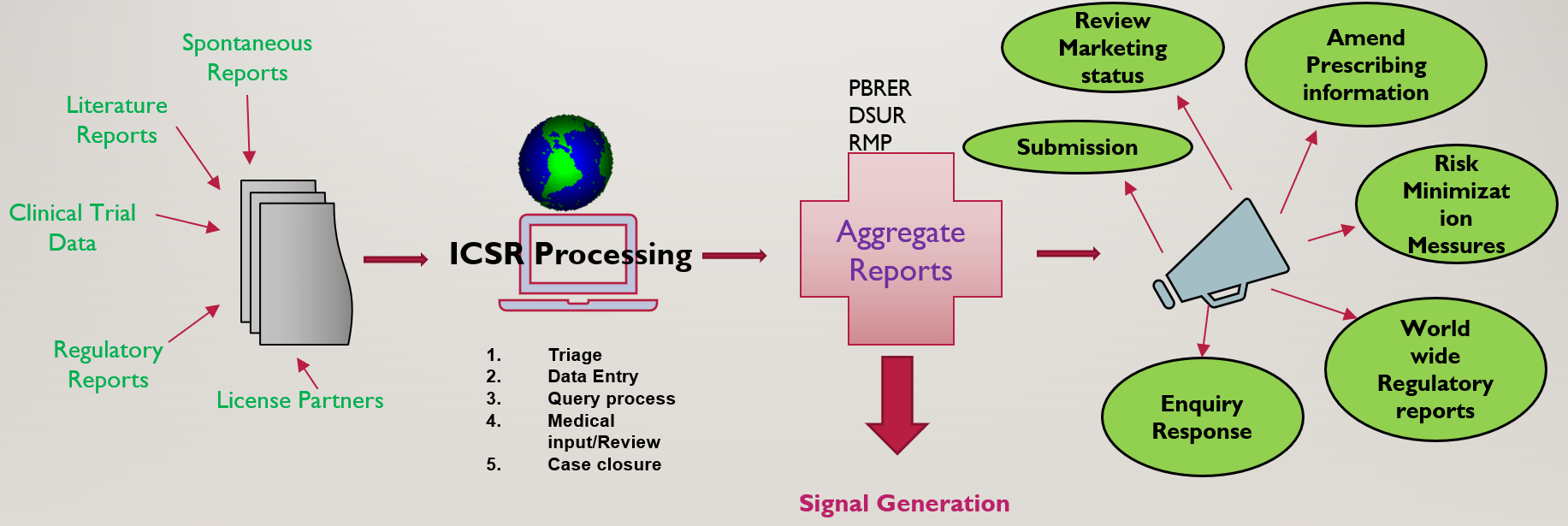
![PDF] Development of a pharmacovigilance system in a resource-limited country: the experience of the Democratic Republic of Congo | Semantic Scholar PDF] Development of a pharmacovigilance system in a resource-limited country: the experience of the Democratic Republic of Congo | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8ad20883b2ad040f9cc202658b049f39b7e53de5/3-Figure1-1.png)
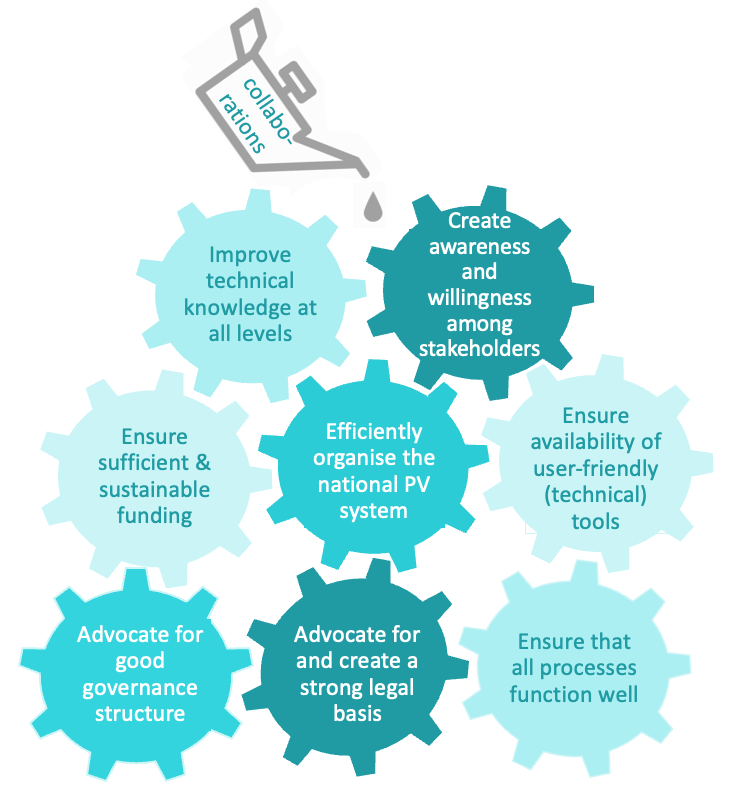
PDF] Development of a pharmacovigilance system in a resource-limited country: the experience of the Democratic Republic of Congo | Semantic Scholar
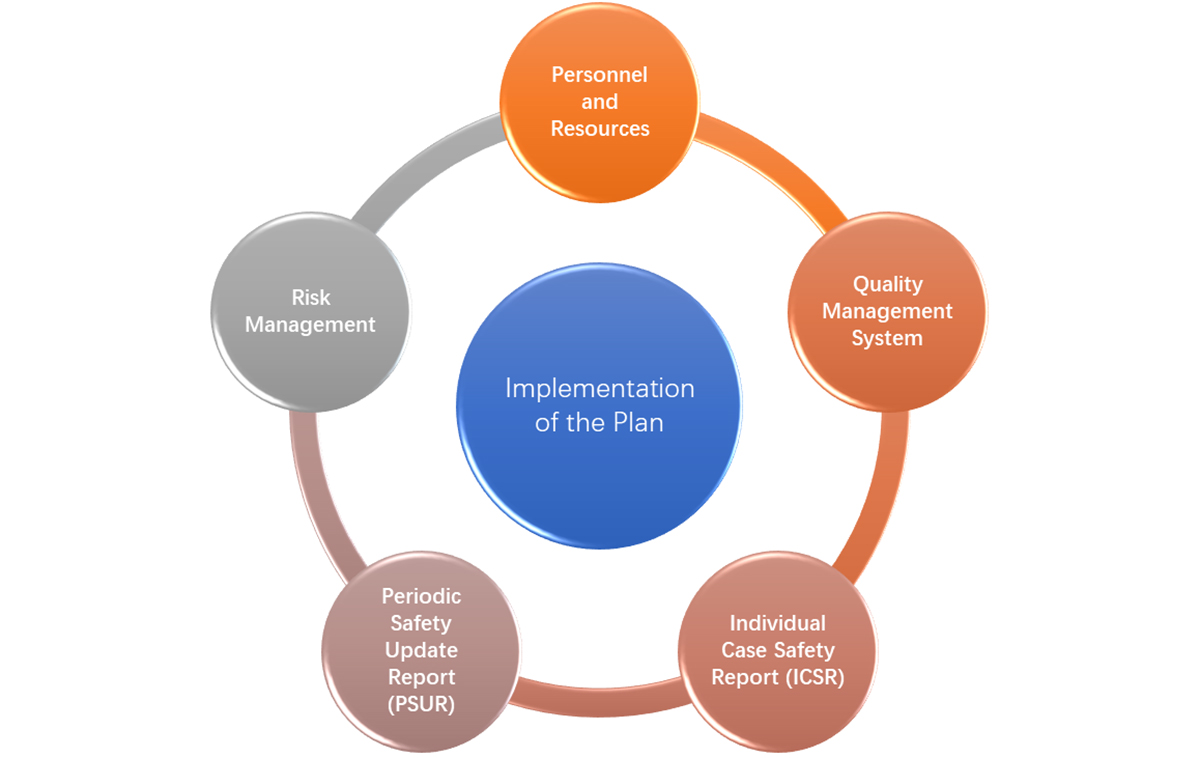
How Global Pharma Companies Implement Good Pharmacovigilance Practices (GVP) In China - Accestra Consulting