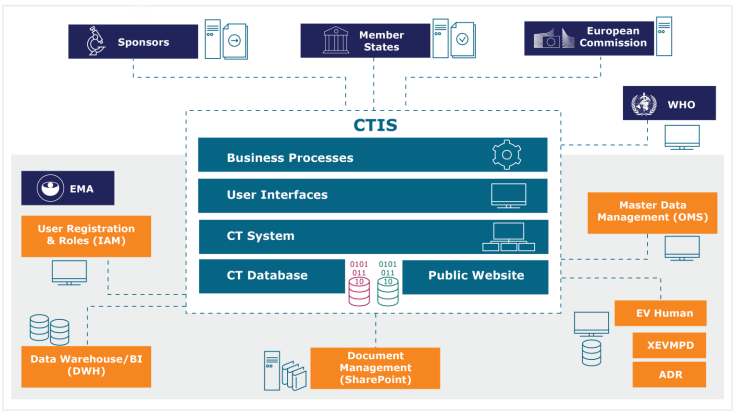
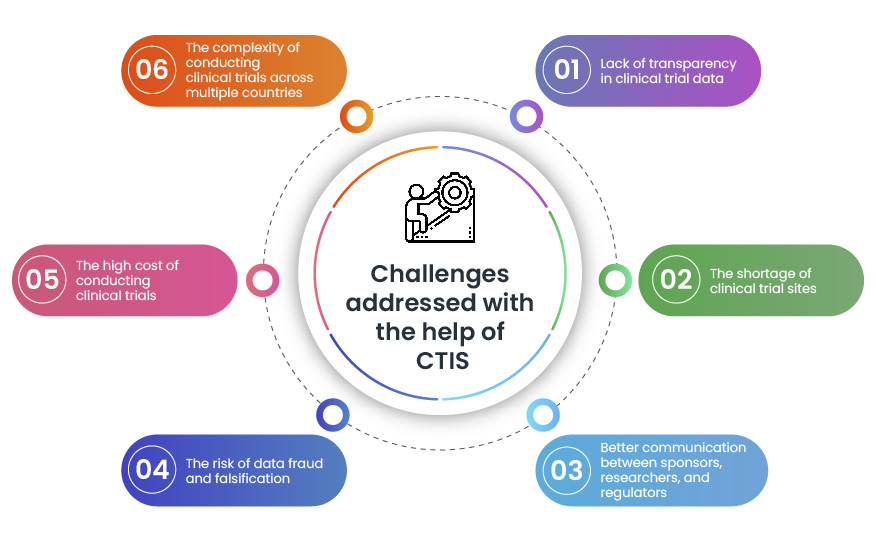
Managing the New EU Clinical Trials Regulation 536/2014 – Guidance for Navigating the Clinical Trial Information System (CTIS)

EMA Webinar for SMEs and Academia on the Clinical Trials Regulation and the Clinical Trials Information System | ERICA

CTIS – M07 How to create a CT: Clinical Trial centric approach vs organisation centric approach - YouTube
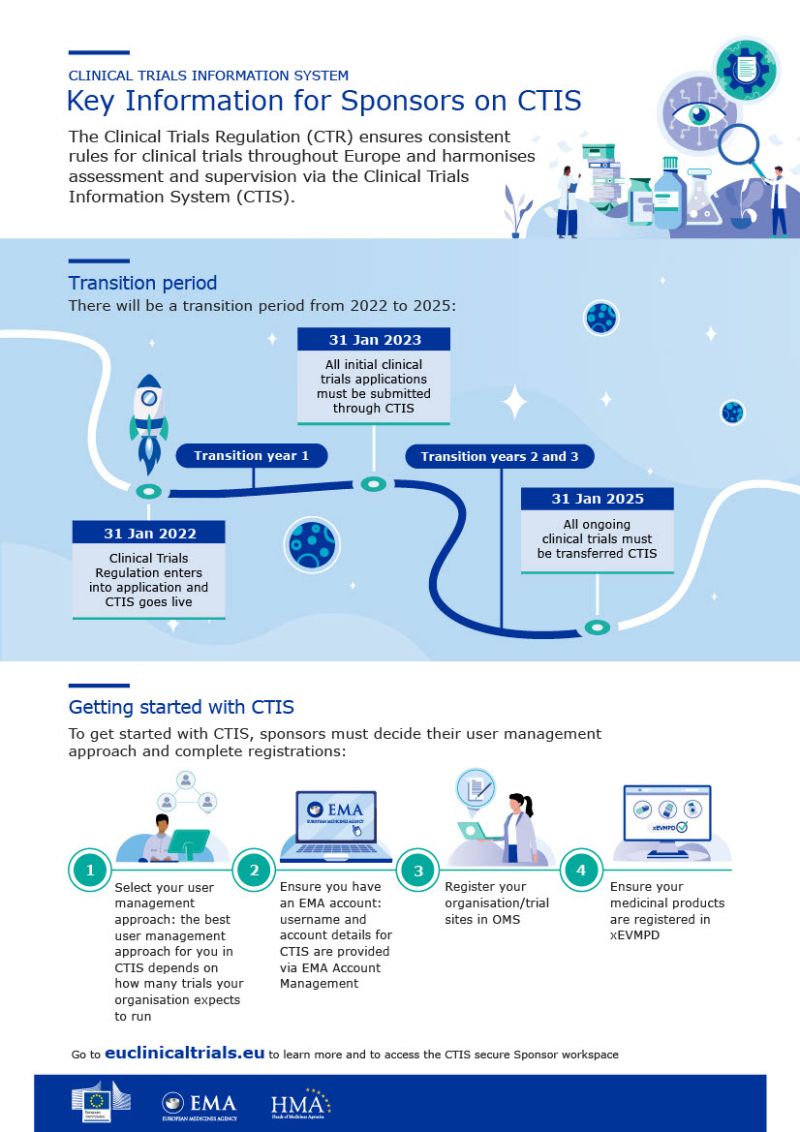
European Medicines Agency na LinkedIn: #euclinicaltrials #clinicaltrials #clinicaltrialsmanager | Komentářů: 22

ICTD 2021 - EMA Clinical Trial Information System (CTIS) for platform trials - Noémie Manent | Ecrin